Real-Life Math -- Solution
By your calculations, how much water vapor is in the hydrogen? How
does it compare with the readings from the humidifier?
Use the formula:
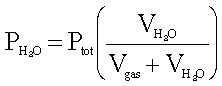
Where:
PH20
= 7.1 PSIA
Ptot = 44.7 PSIA
Vgas = 28 SLPM
Put
the values into the formula.
| 7.1 = 44.7 x | VH20
(28 + VH20) |
Move 44.7 to the left side of the equation
Move the denominator (28 + VH20) to the left
side
0.1588 x (28 + VH20) = VH20
Multiply
it through
4.447 + 0.1588 VH20 = VH20
Keep
working the equation:
4.447 = VH20 - 0.1588VH20
4.447
= VH20(1 - 0.1588)
4.447 = VH20
x 0.84
VH20 = 5.29
You've determined that
the volume of water vapor in the hydrogen is 5.29 SLPM. Yet the humidifier
is still reading 12 SLPM. Obviously, the humidifier's readings are wrong.
First, you'll check your calculations. Then, you'll try recalibrating (adjusting)
the machine's instruments to see if that will solve the problem. If it doesn't,
you may have to order a new machine.
Part of a chemical technician's
job is to keep equipment operating properly, says Neil Fink. If you don't,
you'll get useless data. You'll have to start all over again.